|
Encouraging results using a sulfide-specific electrode were reported. The problem with this approach is the correct sulfide dosage. Sulfide can be added in soluble form as Na2S or NaHS. Because H2S is toxic, the workplace H2S limit is set by the Occupational Safety and Health Administration (OSHA) at 10 ppm. The odor detection level of hydrogen sulfide is 0.1- 1 ppm. In practice, processes are designed to operate with lower sulfide concentrations in order to minimize the H2S odor in the working environment. The most soluble sulfide (MnS) listed in Table 3 would shown an equilibrium concentration of = Kspl 10-7 = 10~7 mol/L = 5.5 mg/L under these precipitation conditions. H2S-* 2H+ + S2' K3 = 10"19 8 (mol//)2 (27)įrom Equation (27), we calculate that at pH 7 a hydrogen sulfide solution of 0.1 mol/L contains only 10-7 mol/L of free S2_ ions. HS~ S2~ + H+ K2 = 10"12 9 molII (26) We find for the overall reaction: The solubility of H2S in water is 0.1 mol/L. Sulfide (S2_) is the dissociation product of H2S. (o) Solubilities of metals precipitated with ferrous sulfate-NaOH from a solution containing 20 ppm each of CrVI, Pb, Cu, Cd, Zn, and Ni as a function of pH. (-) Solubilities of metals precipitated with NaOH from single-metal solutions as a function of pH. Precipitation pHįigure 2 Metal removal efficiency by hydroxide precipitation and Fem coprecipitation. Table 3 Solubility Products (KSJ>) for Metal Sulfides of Type MeS, Me2S, and Me2S3 MeS K Me2S K,p Me2S3 Ksp Metal sulfide precipitation occurs if the solubility product is smaller than the product of metal concentration times sulfide ion concentration: Table 3 lists the solubility products for metal sulfides of the type MeS, Me2S, and Me2S3. Also, the pH range for the precipitation of metals as sulfides is much broader. This should make it feasible to precipitate the heavy metals from solutions containing complexing agents that prevent metal removal by hydroxide precipitation. 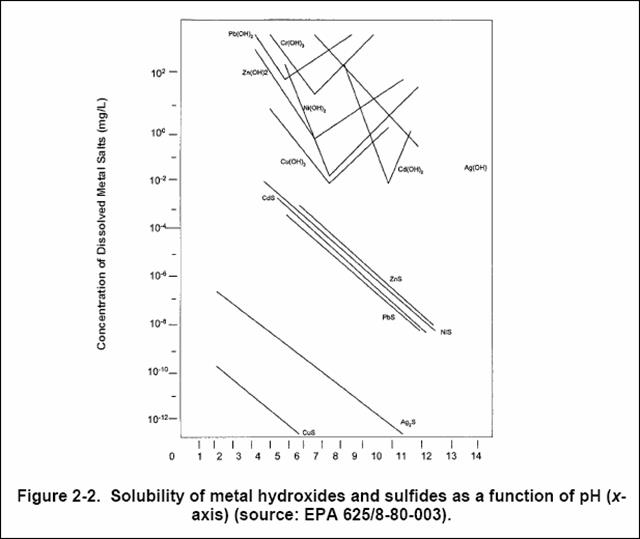
The solubility products of metal sulfides are several orders of magnitude lower than those of the corresponding hydroxides.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
